|
|||||||||
|
|||||||||
|
Green Lead MCA speech Notes for the PowerPoint
presentation |
|||||||||
|
About Us Visitor
Number |
|||||||||
|
"GREEN LEAD" – OXYMORON OR FUTURE VISION? Elizabeth O’Briena,
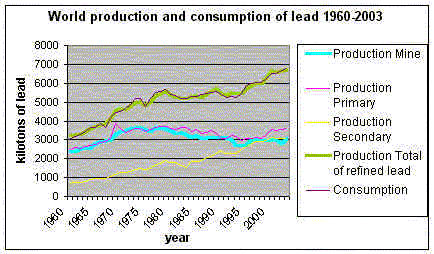
Cornelia Dostb, Bei Quc a Manager, b,c Interns, Contents BATTERIES "100% recyclable" The Battery Council International claims: "the recycling rate for the years 1999 – 2003 is 99.2%." "Compared to 55% of aluminum soft drink and beer cans, 45% of newspapers, 26% of glass bottles and 26% of tires, lead-acid batteries top the list of the most highly recycled consumer product" (Adam and Davidson, 2001). According to that figure one might believe that the demand for primary lead should decrease but Figure 3 below shows a slightly different development. Figure 3 makes clear that the production of secondary lead has risen in the past few years whereas the production of primary lead has been relatively steady and the production of mined lead declined only marginally. The world market for lead is about: 6.8 million tons per annum. The International Lead and Zinc Study Group (ILZSG) estimates that 77.4% of this total amount went into batteries - equivalent to 5.26 million tons. World mine production in 2003 was 3.1 million tons. Western world secondary lead production represented 65% of total refined metal production (Burrell, 2005). Figure 3
Why is there still a steady demand for primary lead? The reasons could be as follows: Lower costs of producing primary lead According to the Lead Development Association International the main disincentives of recycling lead are low costs of raw materials to produce virgin lead and fairly high collection and transportation costs for a low value product. "Some lead products are not recycled, either because it is not economic to do so at present, or simply because it is not practical to do so." (Imperial College Consultants Ltd., 2001). 65% recovery rate from used lead acid batteries According to Dr Peter Hurley of Blake International Limited, an OH&S consultancy in the UK, battery makers can only use two thirds (65%) of what is recovered from used lead acid batteries. "Even in the US, much of the recovered lead is exported. The reason this is so is because they use lead-antimony alloy, but cannot use that alloy in the anode plate of the battery as its too corrosive in that application. If they used lead-tin alloys then they could reuse nearly all the material they recovered/recycled, provided they could keep it separate from the lead-antimony. But they don't, because they can't. Face it, when they're recycling batteries, who discriminates between a calcium, selenium, tin or antimony alloy battery? And how can any recyclers, let alone consumers tell the difference? "A lead-tin battery will last up to five times longer than a conventional lead-antimony battery, and the battery maker can't get 5 times the profit from the better battery. The greatest part of the battery market is for replacement batteries, as these batteries are sold via auto-factors to the users it makes the battery maker the most profit per unit, and it would be this sector that would be impacted the most. Sales to car-makers are big volume but low unit profit. Switching to lead-tin could potentially cause a 90% reduction in the profitability of the battery makers if the car-makers would not pay more. "Also the consequence of all that efficient recycling could cause a minimum of 70% reduction in demand for fresh mined lead. Bad news if you're a mining company. Face it how many mining companies put the environment ahead of profit in their thinking. So the big question is; with such a big environmental advantage to be gained from this technical switch, will the battery industry move away from lead-antimony voluntarily? Answer - Some niche makers have, others want to but some big players won't play ball. Unless they all do so the secondary lead feed-stock will continue to be contaminated by antimony and 1.1 million tonne lead alloy will still go missing every year. I would very much like to know where it goes missing to. If anyone has the answer let me know." (Peter Hurley, 2005a) Large-scale international marketing Since more and more lead is exported to overseas markets, this also increases the difficulty for local battery companies to collect and recycle all the batteries they produce. In 2004 Australia was the largest exporter of lead ore and concentrates, and the second largest exporter of refined lead. In the last financial year (2003/04) Australia produced 247 kilotons of primary lead and only 35 kilotons of secondary lead. Furthermore, 231 kilotons of refined lead (including primary and secondary lead) have been exported. The consumption of refined lead (primary and secondary lead) has been up to 60 kilotons in Australia only (Australian Commodity Statistics, 2004). Put succinctly by Dr Peter Hurley, "The world market for lead is about: 6.2 million tonnes. There is 3 million produced from mining each year. The car battery makers consume 3.2 million tonne recycled lead and 1.4 million tonne new mined lead, which they need because the maximum they can use is 65% secondary recycled lead (due to anodic corrosion). Of the 4.6 million tonnes used to make batteries every year world-wide only 3.5 million tonnes gets recycled. My problem is I don't know where the other 1.1 million tonnes goes." (Hurley, 2005b) Increased Demand and Consumption Recently, lead acid battery consumption within OECD countries continues to rise and even more in the developing countries with fast growing industrial nations like China. Many of these countries do not have any or enough domestic supply. Asia is experiencing a large growth in the automobile market. This means the demand for batteries therefore also increases. Up to now this incredible demand for lead cannot be satisfied by secondary lead only. In addition, the growing market for batteries creates a lag of 4-5 years between use and recycling. Thus, the imports of primary lead increase as well as the demand for scrap material. Moreover, consumers of lead acid batteries in many countries do not keep records of sales and recycling rates and therefore nobody seems to know the exact figures for the amount of lead that is circulating worldwide. However, it will take a while before the statistics are complete as many of the recyclers in the developing world operate without proper licenses or "informally" and therefore do not provide Government agencies with recycling tonnages (Burrell, 2005). Besides those problems that seem to hinder the recycling rate of lead from reaching and exceeding 100%, there is still the fact that a hazardous product cannot be recycled 100%! "Just as the primary plastics industry promoted plastics "recycling" when citizens in industrial countries began fighting for plastics packaging bans, the lead-acid battery industry is using the cloak of "recycling" to hide the impact of its products' wastes, and to thus reduce the threat to its 'status quo' use of toxics in production processes" as fittingly stated by the authors of Greenpeace article ‘The Myth of Automobile Battery Recycling’ (Cobbing and Divecha, 2005). For now it seems that "green lead", even when applied only to the lead in batteries, will remain an oxymoron and it will be difficult and challenging to reach and exceed 100% recycling rates. 100% would be the recycling rate if all the batteries that were made in one year were recycled, but because so much lead is "out there" the rate of recycling must be greater than 100% for many years to come, in order to bring back all the batteries not recycled in their year of manufacture – globally this is at least 35% of batteries for many decades. That means the lead problem will continue to exist for a very long time and consequently lead poisoning and environmental degradation goes on, especially in many developing countries. Contents |
|||||||||
|
About
Us |
bell
system lead poisoning |
Contact Us
| Council
LEAD Project | egroups | Library
- Fact Sheets | Home
Page | Media Releases |
|||||||||
|
Last Updated 4 March 2026
|
|||||||||